Briefing you on the latest in reproductive health research
TL/DR: This month’s update discusses a new federal initiative focused on addressing maternal mortality rates in the U.S., along with a newly pre-qualified medication delivery system that may feel familiar – but this time for STI prevention. Plus, we discuss pregnancy for breast cancer survivors and a doctor role model for us all. Here’s the latest.
Your tax dollars at work
The U.S. Department of Health and Human Services (HHS) announced last week an Action Plan devoted to addressing rates of maternal mortality, or death of pregnant people, in the United States.1
The main goals of the plan are to reduce the national maternal mortality rate by 50%, reduce the C-section rate by 25% for low-risk pregnancies, and ensure controlled blood pressure in 80% of reproductive women with pre-existing hypertension… all by 2025. This is a lot to digest, so let’s break it down.
Maternal mortality rates:
This refers to the number of pregnant women that die during pregnancy or as a result of pregnancy-related consequences. This number is about 700 in the United States per year. Importantly – and tragically – Black and indigenous women are 2-3 times more likely to die from a pregnancy-related cause when compared to white women.2 This is a matter that deserves urgent attention and action.
C-section rates:
This is a topic for a much longer conversation, but there is much debate in the literature about C-section rates — when to do them, and for whom, and for what reason. Without going into too much detail, C-sections do represent a more invasive surgery and thus have more risk, which is likely why this plan proposes reducing C-section rates for low-risk pregnancies nationwide.
Blood pressure control during pregnancy:
Again, a topic for a much longer discussion – but in short, having uncontrolled or high blood pressure during pregnancy can put the pregnant person at risk for a number of complications to both mother and child later on. Helping the pregnant person with their cardiovascular or heart health early on can be beneficial later down the line.
So far, the government has released this document alongside tentative plans for a public-private partnership to achieve these aims, so we will see how this progresses in the coming months.
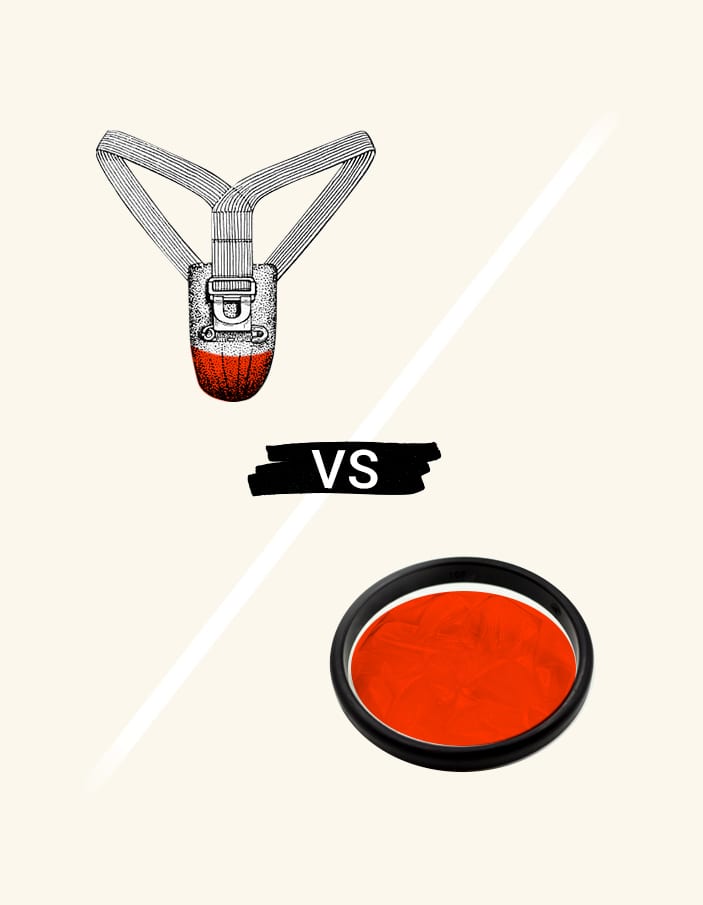
Dapivirine vaginal ring is pre-qualified for HIV prevention by the WHO
The vaginal ring, more commonly known as “the ring,” is best known for its role as a contraceptive option. Vaginal contraceptive rings approved for use in the U.S. function by releasing hormones such as estrogen into the vagina locally, where they are quickly absorbed into the bloodstream. This prevents pregnancy via hormonal regulation.
For years, researchers in sexually transmitted infection (STI) prevention have brainstormed about innovative ways to deliver treatment – and the vaginal ring became a source of inspiration when, in 2016, those researchers began to consider a more direct approach to delivering preventative HIV medication.
Rather than supplying synthetic hormones for pregnancy prevention, the dapivirine ring aims to prevent HIV acquisition (the process of becoming infected) by delivering medication directly to the area in which transmission would typically occur. The medication then prevents viral replication, a.k.a. the virus copying itself.
Early scientific studies of the dapivirine ring were promising, showing that consistent use of the ring reduced the risk of HIV infection by about 30% for women aged 18-45.3 4 The authors also concluded that there weren’t any concerns around safety, strengthening its potential for use.
Dapivirine ring is co-signed by important health authorities
Earlier this year, the European Medicines Agency (EMA) concluded that it had a positive opinion on the dapivirine ring, an encouraging milestone in the ongoing fight against HIV and AIDS.5 Even more recently, on World AIDS Day which fell on December 1st, it was announced that the World Health Organization (WHO) had officially pre-qualified the dapivirine ring for HIV prevention use.6
In this setting, pre-qualification means that this novel medication would be directed towards areas that have the highest burden of HIV. However, following the appropriate regulatory processes, the dapivirine ring could one day be available in the U.S. and in other countries, as well, serving a critical role in the expansion of STI prevention therapies. This is one to watch in the coming years.
Fertility preservation for breast cancer survivors
In other news, a recent analysis published in JAMA Oncology provides some optimistic insights on navigating pregnancy following a diagnosis of breast cancer.7 Fertility preservation has been a hot topic in the field for quite some time, and there is a body of research emerging that focuses on fertility in women who have a history of cancer in one of the reproductive organs, such as the breast, uterus, cervix, or ovaries.
Fertility preservation procedures that remove and store oocytes – an immature, unfertilized egg cell – can be painful, expensive, and physically and psychologically taxing, which is why it is so important to make the decision to undergo this procedure with the guidance of a compassionate and skilled OB-GYN.
The JAMA Oncology study, which was conducted across a Swedish population, analyzed the data of female-identified patients who did and did not choose to undergo fertility preservation after a diagnosis of breast cancer. The researchers followed these women for over 20 years in order to see the success of subsequent pregnancy in both populations.
Fortunately, both cohorts of women (those who elected for oocyte retrieval and those who did not) were able to become pregnant in the future, with those who had done fertility preservation having higher rates of births.
Importantly, the researchers did not find any association with all-cause survival, which is a scientific way to measure how many patients stay alive after a diagnosis. This goes to show that deciding to undergo fertility preservation procedures did not significantly influence survival, which will be so helpful for making those decisions during the time of a difficult diagnosis – especially considering the fact that breast cancer can affect women of reproductive age.
COVID-19 leadership driven by engagement on health equity
In the past couple of weeks, there’s been news of several appointments to offices critical for health reform in the U.S. These appointments will have an impact on sexual and reproductive health initiatives at the federal, state, and local level. Of these, several female leaders have taken center stage:
Dr. Marcella Nunez-Smith has officially been named the COVID-19 Equity Task Force Chair for President-elect Biden. Dr. Nunez-Smith is a celebrated scholar, academic, and physician who currently works on health equity research with a focus on structurally marginalized populations at the Yale School of Medicine. We applaud Dr. Nunez-Smith and we’re excited to witness her impact as the battle against COVID-19 continues on.
Here is a quote from a recent interview that exemplifies her leadership:
“The pandemic, very sadly and unfortunately, laid bare what were pre-existing structural and social realities that really predisposed particular communities to be hardest hit by this pandemic…We’ve had a collective witnessing as a country here in 2020 around the pervasive, deep-seated challenge of racial injustice, and Covid-19 exploited that reality.” 8
Addressing health disparities is fundamental to all parts of public health, from infectious diseases like COVID-19 to reproductive conditions like endometriosis. Knowing we have an expert with a diverse background and progressive mission at the helm is a sign of positive change to come.
This article is informational only and is not offered as medical advice, nor does it substitute for a consultation with your physician. If you have any gynecological/medical concerns or conditions, please consult your physician.
© 2021 The Flex Company. All Rights Reserved.